 
However, at the thermodynamic limit ( $N\rightarrow \infty$), terms finite or proportional to $\ln N$ do not contribute to the macroscopic entropy. For entropy of mixing to exist, the putatively mixing molecular species must be chemically or physically detectably distinct. 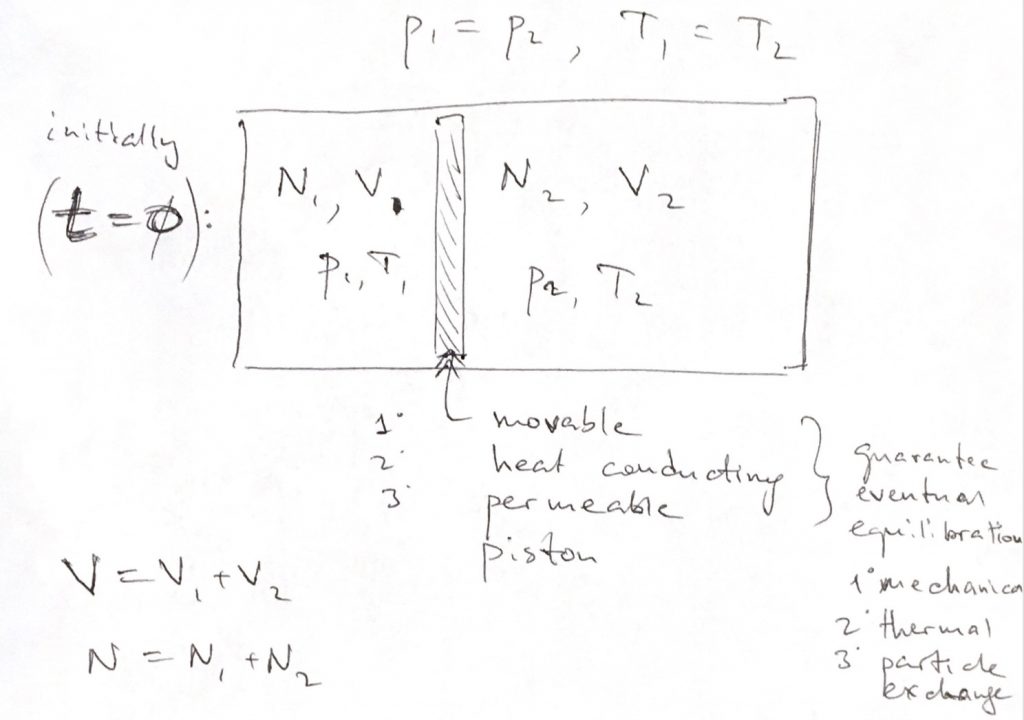
The logarithm of such an expression multiplied by the Boltzmann constant is the entropy of mixing, and it is clearly positive. The change of entropy with respect to a system made by two equal subsystems of $N/2$ particles, each with $P/2$ states, can be obtained by taking the logarithm of the ratio $\frac. Indeed, let's indicate by $F(N,P)$ the number of microstates of $N$ identical particles which can be in $P$ different states. In that limit the additional states do not contribute. The reason is that thermodynamic entropy can be obtained from statistical mechanics only in the limit of large systems (the so-called thermodynamic limit). Deducing general formula for information entropy of mixing molecules and its. The weights may be interpreted as the probabilities to. The volume fractions of the two components in the mixture are thus given by (7.1.1) A V A V A + V B (7.1.2) B V B V A + V B 1 A. Information entropy h (or Shannon entropy) of the molecule is introduced as a sum of the logarithms of the weights of each atom type (N j / N j): h - j 1 n N j j 1 n N j log 2 N j j 1 n N j. However, these additional states do not contribute to an increase in the macroscopic thermodynamic entropy. Entropy of Binary Mixing We consider mixing of two species A with volume V A and B with volume V B and neglect volume change, so that the total volume is V A + V B. In total, $10$ states to be compared with the $4$ states in the presence of the divider. Ram Nandan Singh 1, Victor Witusiewicz Add to Mendeley Cite (01)01272-5 Get rights and content Abstract The excess entropy of formation of binary alloys has been described as the sum of configurational, vibrational, electronic and magnetic contributions. If we do it slowly enough we will have reversibly unmixed the two gases by something similar to reverse osmosis, using an amount of work equal to the minimum $W$ calculated above.The situation is even worse in the case of non-interacting particles: there are four additional states due to the possibility of occupying a single slot with two particles. We can now very slowly push the two pistons in opposite directions until they meet in the middle, while holding the system in contact with a heat bath. One is permeable to the first gas but completely blocks the second, and the other is permeable to the second gas but blocks the first. S m i x r is expected to be vibrational to a large extent, but not entirely so. The former is the entropy of mixing accessible to the liquid, the latter is the mixing entropy in the vitrified mixture. It's possible for this minimum bound to be negative, meaning that you can actually get work out of the system by unmixing the fluids - but in this case the fluids would be immiscible, so the mixed state would be the unstable one.įinally, let's make the additional assumption that the fluids are gases and that we have two very special semi-permeable pistons to hand, one at either end of the container. The entropy of mixing consists of two parts, a configurational portion ( S m i x c), and a residual part ( S m i x r). In this case (and only in this case), the minimum work required is equal to the difference in Helmholtz free energy between the mixed and unmixed states. In practice you'd usually assume the fluids to be in contact with the heat bath, so that the two temperatures are in fact equal. To define the solvus, the use of a configurational excess entropy is needed in most cases, which is, however, not listed because it is only. I didn't need to make any assumptions about the fluids' temperature in order to work out the above. Ideal mixing is defined in the last column (a id ). Without the heat bath there would be nowhere for the energy from the work source to go once it's used up.Īlso note that $T$ is not necessarily the temperature of the mixed fluids, which could be different from that of the heat bath. 
But note that the expression involves $T$, which is the temperature of the heat bath that I assumed to exist. What we know for sure is that the entropy of the universe as a whole cannot decrease, and hence fluids can't be unmixed without increasing the entropy of some other system by an amount equal to $\Delta S_\text + \Delta H.$Ĭhanging this to an equality gives you the minimum amount of work required to unmix the fluids. This question can be answered, but only with the addition of some extra assumptions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
